2 min read
Some severe inflammatory intestinal diseases remain resistant to standard treatments. What if the root cause lies in a fundamental dysregulation of the immune system?
Hypothesis
The JAK-STAT pathway is a central driver of inflammation. It is normally controlled by molecular “brakes”, including SOCS1. If this brake fails, immune activation can become excessive and persistent.
What we did
We studied two adult patients presenting with very atypical intestinal diseases:
- one with treatment-resistant Crohn’s-like inflammation
- one with a rare and severe condition called chronic intestinal pseudo-obstruction
Using genetic sequencing, we identified in both patients a mutation affecting SOCS1.
What this changes
This finding connects:
- a specific genetic defect
- to a well-defined biological mechanism (JAK-STAT hyperactivation)
- and to distinct clinical diseases
The therapeutic impact
Instead of relying on trial-and-error treatments, we used targeted therapies:
- IL-12/23 blockade
- JAK inhibition
This led to a marked clinical improvement, including in both cases.
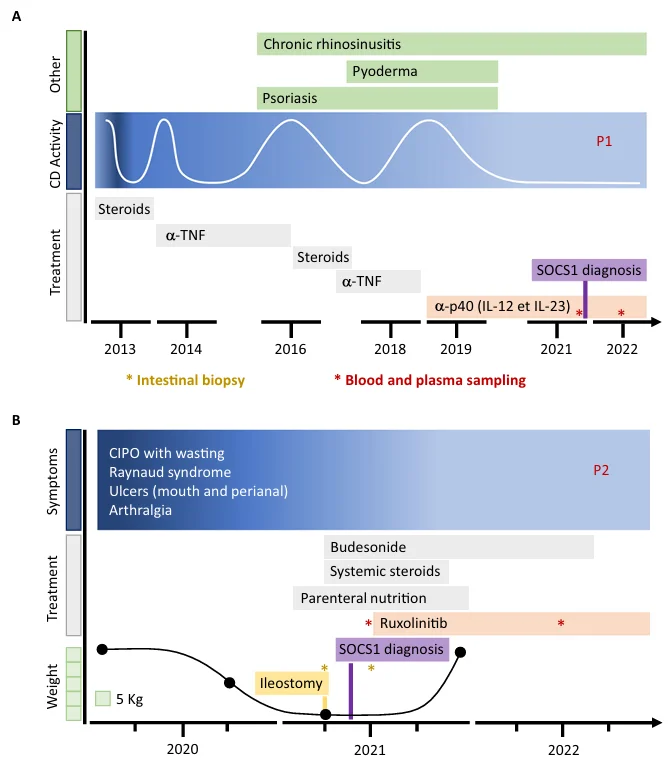
Disease timeline and treatment strategies
A — P1 was initially treated by infliximab (anti-TNF-α) and after relapse by ustekinumab (anti-IL-12/IL-23). B — P2 was initially treated with systemic steroids. Ruxolitinib was introduced after SOCS1 diagnosis. A single asterisk (*) indicates a blood sampling before and after 1 year of treatment.
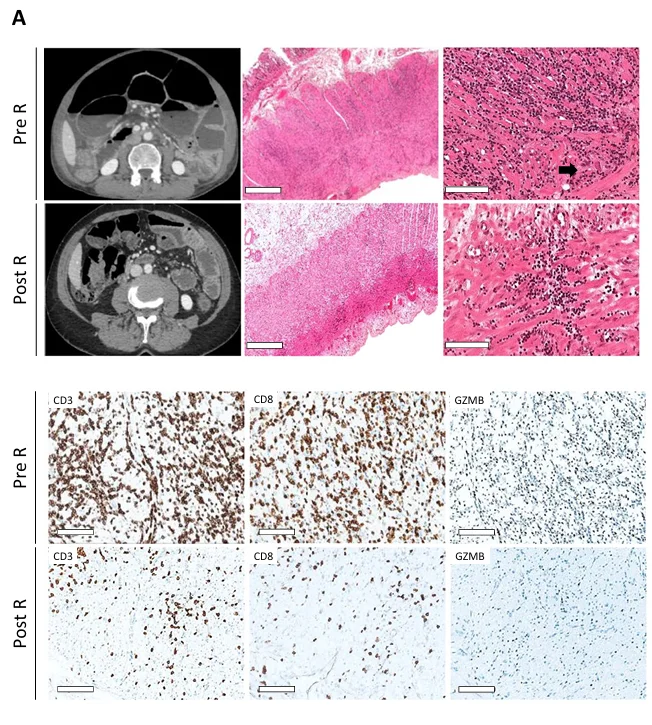
Histological resolution under ruxolitinib — P2
CT scan, histology of lymphocytic intestinal leiomyositis in surgical specimens HES staining (scale bar = 900 μm or 100 μm) and immunohistochemistry staining of CD3+, CD8+, GzB+ lymphocytes before and after 3 months of ruxolitinib (R) (scale bar = 100 μm).
Why it matters
- Shows that a single genetic cause can lead to distinct clinical phenotypes
- Supports the use of genetic testing in atypical or treatment-resistant cases
- Provides evidence for the effectiveness of targeted therapies
Other publications

Histogenetic Classification Predicts Outcomes in 130 Adults With Chronic Intestinal Pseudo-Obstruction
Integrating genomic and histopathological data resolved 82% of idiopathic cases. Monogenic myopathy emerged as an independent predictor of favorable long-term survival.

Late-Onset Status Epilepticus Associated With Isolated Leptomeningeal Angioma and Sturge-Weber Syndrome-Related GNA11 Pathogenic Variation
Some unexplained adult-onset epilepsies may have an underlying vascular-genetic origin.
NR1D1::MAML3 Fusion in an Aggressive Mesenchymal Neoplasm
NR1D1-rearranged tumors are emerging aggressive mesenchymal neoplasms.

PFMG2025-integrating genomic medicine into the national healthcare system in France
The 2025 French Genomic Medicine Initiative represents one of the first nationwide efforts to integrate whole genome sequencing into routine clinical care.